You may have thought you were done with organic chemistry after your college undergraduate classes but—surprise!—you will see it on the MCAT.
The three big questions about organic chemistry on the MCAT are:
Organic chemistry content will appear in two sections on the MCAT.
Only about 12 of the 230 MCAT questions are directly based on organic chemistry. However, a good foundation in organic chemistry principles will not only help you with those questions but with navigating complex biological structures and deciphering biochemical processes across the entire test.
Note : You will rarely be required to go into the level of detail that you did in your undergraduate organic chemistry classes.
First, let’s talk about how you will be asked to apply your organic chemistry knowledge on the MCAT.
You will NOT need to perfectly memorize and regurgitate reaction mechanisms or conjure up intricate reaction products after following an endless reaction scheme.
You WILL need to rely on a solid foundation in the fundamentals along with an ability to quickly relate that knowledge to information presented inside a passage or a question.
The MCAT is known for layering subjects so that you have to apply reasoning across closely related concepts. In this case, you will need to demonstrate an understanding of organic chemistry concepts and principles in relation to biochemical processes and structures (as seen in the sample questions at the end of this article). The MCAT is testing your ability to locate important information within a passage, reaction, or structure.
Biological structures are a vast subject area, but don't let that intimidate or distract you. Instead, use practice problems to practice focusing your attention and quickly reading paragraphs to extract the important details. This will also improve your content background.
If you’ve taken a two-semester course in organic chemistry, then you’ve likely seen more than what the MCAT will test. It’s important to know what you’re responsible for studying.
Here are the five broad categories of organic chemistry content you will need to focus on:
The concept of stability is a major one in organic chemistry, and it relates to many other concepts that will be tested. The golden rule you must know is that stability is inversely related to reactivity. This means that the more stable a substance is, the less reactive it will be.
Your ability to predict the stability of a molecule will be based on identifying the presence of stabilizing principles like:
as well as destabilizing influences like:
For example, a charge within a molecule often epitomizes instability, but if that molecule possesses the ability to mitigate that charge, perhaps via resonance, it may become sufficiently stable. This is the foundation for predicting important reactivities in organic compounds like:
These details will relate more directly to your ability to learn and identify organic reactions that may appear on the MCAT.
Being familiar with functional groups in organic molecules is an integral prerequisite to being able to examine a structure and identify its most important features.
The MCAT prioritizes the following compounds above others, as these are most likely to appear within biological molecules:
You will also need to be familiar with nomenclature for those functional groups and their properties, which will pay off when learning the reactions they participate in.
Isomers are the various molecular structures that may be created from a single molecular formula. It is important to learn the various subdivisions found within this umbrella term, which is where details emerge.
Test-takers must be familiar with three major isomer types:
It is also important to be comfortable doing the following:
There are two classes of laboratory techniques that may be tested concerning organic molecules:
For each technique, it is crucial to know the basis for separation (e.g., polarity, charge, size) and the order separation is expected to occur in.
For each technique, it is imperative that you know what information each form of spectroscopy provides about a molecular structure and that you know common characteristic absorptions.
Note: In most cases, undergraduate organic chemistry classes go much deeper than the MCAT will when testing content from this area.
Organic Reactions can be split into three major categories on the MCAT:
Many reactions touch on carbonyl chemistry, which is one of the most important functional groups in biology and therefore likely to be tested.
Other important organic reactions include redox reactions and acid-base reactions , largely as they pertain to alcohols and carbonyl-containing compounds.
MCAT test-takers consistently say the same thing: know your amino acids ! Amino acids are aggressively tested on the MCAT, especially in the context of biochemistry, so it’s important to be very familiar with their details.
For all 20 canonical amino acids, you will need to know:
You must also be able to apply these details within the context of peptides and proteins. For example, you may be asked to determine the overall charge on a peptide in solution, or to predict the consequence of an amino acid site-mutation in the function of a protein. Scroll down for examples of some amino acid-based practice problems.
From the standpoint of structure and reactivity, familiarity with the following is necessary:
The key to success in organic chemistry content on the MCAT is to build a solid foundation in the fundamentals. Exposure to organic chemistry practice problems is a necessary part of this foundation that takes time and can’t be rushed. Over time, you will become more comfortable examining molecular structures for their vital parts, which is routine for organic structures presented on the MCAT.
One last thing to keep in mind: as your grasp of organic chemistry concepts grow stronger, so will your grasp of biochemistry concepts and processes, so there is a significant payoff for the time you invest in prepping organic chemistry for the MCAT beyond what you might expect.
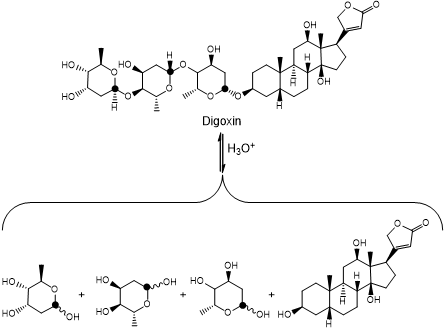
How many fragments are expected to result via acid-catalyzed hydrolysis of digoxin?
Explore our featured graduate schools & programs to find those that both match your interests and are looking for students like you.
Check out our complete list of 168 law schools, based on surveys of school administrators and over 17,000 students.
Visit our Med School Hub to explore med schools with our ‘Find Your Med School’ filtered search or visit our Med School Advice pages for info about good MCAT scores or interview question prep.

Explore our featured business schools to find those that are looking for students like you.